What is Daucus carota?
Daucus carota, commonly known as the carrot, is a versatile dicotyledonous plant belonging to the Apiaceae family. The primary edible portion of the carrot is its root. It is renowned for its rich content of vitamins A and C, dietary fiber, and an array of antioxidants. Carrots are globally cultivated, serving as a culinary ingredient and a source of animal feed.
Daucus carota metabolomics analysis is a powerful scientific approach that delves into the metabolites of carrots. Metabolites are the chemical compounds produced within the carrot, including organic acids, amino acids, sugars, enzymes, hormones, and more. These compounds play pivotal roles in vital biological processes such as growth, development, energy production, and responses to environmental stressors.
Our metabolomics analysis services employ cutting-edge instrumentation, including mass spectrometers and nuclear magnetic resonance spectrometers, to detect and quantify these metabolites. Through detailed analysis, we gain insights into the types, quantities, and relative concentrations of metabolites within carrots. This information reveals how carrots respond to environmental factors and growth stages.
Specific Daucus carota (Carrot) Analysis Projects by Creative Proteomics
Phytochemical Profiling: Explore the diverse phytochemical composition of Daucus carota, identifying and quantifying bioactive compounds such as carotenoids, flavonoids, and polyacetylenes.
Nutritional Quality Assessment: Evaluate the nutritional content of carrots, providing insights into their potential health benefits and nutritional value.
Metabolic Pathway Elucidation: Unravel the intricate metabolic pathways involved in carrot growth and development, shedding light on key regulatory mechanisms.
Stress Response Mechanisms: Investigate how Daucus carota responds to environmental stressors, including drought, pests, and diseases, to develop resilient carrot varieties.
Biofortification Studies: Enhance the nutritional profile of carrots through biofortification strategies, aiming to address nutritional deficiencies and improve food security.
Quality Control in Carrot Production: Establish quality control parameters for carrot production and processing to ensure consistency and safety in the carrot supply chain.
Medicinal and Therapeutic Potential: Explore the medicinal and therapeutic potential of Daucus carota, investigating its use in traditional medicine and as a source of bioactive compounds.
Chemical Defense Mechanisms: Study the chemical defense mechanisms in carrots, including antioxidants and antimicrobial compounds, to explore their ability to resist diseases and pests.
Genetic Diversity Research: Analyze metabolic differences among different carrot varieties to understand how genetic diversity impacts nutritional value and resistance.
Food Processing and Preservation: Research carrot processing methods to develop new food products or improve existing processing techniques, extending the shelf life of carrots.
Sustainable Agriculture: Investigate carrot cultivation and management methods to enhance agricultural sustainability, reduce resource waste, and minimize environmental impact.
Breeding Improvement: Utilize metabolomics analysis to identify metabolites associated with specific traits and qualities, expediting carrot breeding improvements.
Ecological Interactions: Study the interactions of carrots with other ecosystem members, such as soil microorganisms, pollinators, and wildlife.
Methods for Carrot Metabolomics Analysis
Sample Preparation: Before analysis, carrot samples must be properly prepared. This typically involves washing, peeling, and homogenizing the carrots to obtain representative samples.
Extraction of Metabolites: Metabolites are extracted from the prepared carrot samples. Solvents like methanol, ethanol, or water are often used for extraction to capture a broad range of metabolites.
Liquid Chromatography-Mass Spectrometry (LC-MS): LC-MS combines liquid chromatography for metabolite separation with mass spectrometry for identification and quantification. Common instrument models include:
- Agilent 1290 Infinity II LC System with Agilent 6550 iFunnel Q-TOF LC/MS.
- Thermo Scientific Vanquish UHPLC System with Thermo Scientific Q Exactive Orbitrap LC/MS.
Gas Chromatography-Mass Spectrometry (GC-MS): GC-MS separates volatile and small polar compounds through gas chromatography and identifies them using mass spectrometry. Common instrument models include:
- Agilent 7890B GC System with Agilent 5977A Mass Selective Detector (MSD).
- Shimadzu GCMS-QP2020 NX Gas Chromatograph Mass Spectrometer.
Nuclear Magnetic Resonance (NMR) Spectroscopy: NMR provides structural information about metabolites non-destructively.
Data Analysis and Bioinformatics: Advanced data analysis tools and software are used for processing and interpretation, including statistical analysis, pathway analysis, and multivariate analysis.
Metabolite Databases: Reference databases aid in metabolite identification.
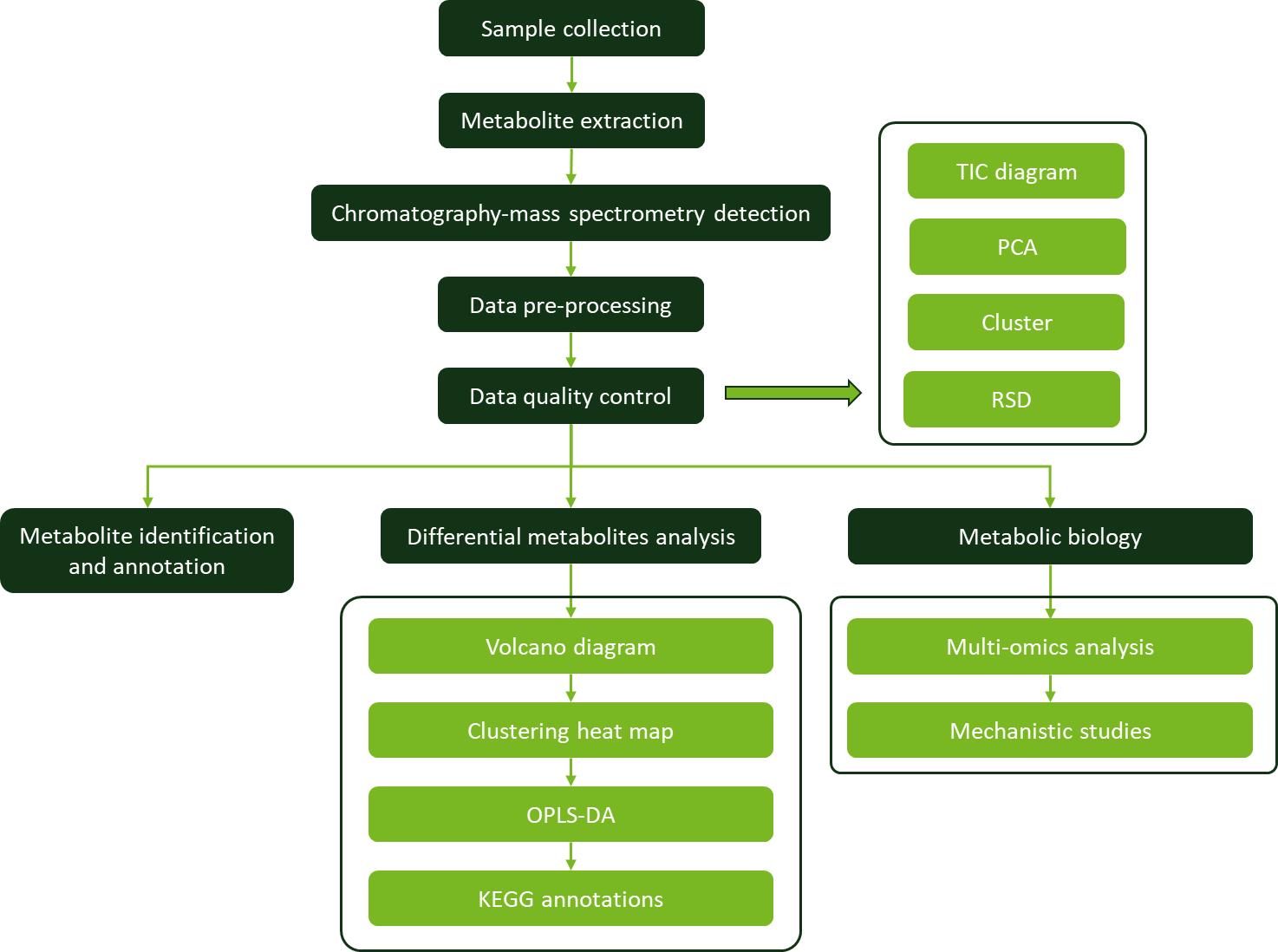 Workflow for Metabolomics Service
Workflow for Metabolomics Service
Sample Requirements for Carrot Metabolomics
| Sample Type |
Recommended Sample Quantity |
| Carrot Roots |
100 grams to 500 grams |
| Carrot Leaves |
50 grams to 200 grams |
| Carrot Stems |
50 grams to 200 grams |
| Carrot Whole Plant |
100 grams to 1 kilogram |
| Carrot Flowers |
20 grams to 100 grams |
| Carrot Fruits |
50 grams to 200 grams |
| Carrot Seeds |
10 grams to 50 grams |
Case. Impact of Farming Conditions on the Quality of Carrots: A Study on Two Cultivars and Three Farming Systems
Background
This study aimed to investigate how different farming conditions affect the quality of carrots, focusing on two carrot cultivars (Afalon and Cortina) and three farming systems (organic, integrated, and conventional). The key parameters of interest were the content of ascorbic acid (AA) and carotenoids, as well as the metabolomic fingerprinting for authentication.
Samples
Carrot samples from the two cultivars, Afalon and Cortina, were grown in experimental plots at the Czech University of Life Sciences in Prague-Troja. The farming systems included organic, integrated, and conventional, with variations in nitrogen fertilization and pesticide use. Different seeding densities (6 × 10^5 and 9 × 10^5 seeds per hectare) were also tested. Each cultivar in every crop system was grown in four replicates, with a total of 12 roots collected per variant.
Technological Methods
Biological Material: Two carrot cultivars, Afalon and Cortina, were used in the study. These cultivars differ in their characteristics, such as vegetation time and suitability for mechanized harvest.
Farming Conditions: The experiments were conducted in Prague-Troja, characterized by dry, medium-warm climatic conditions and a specific soil type (modal fluvisol). Three farming systems were compared: conventional (with specific fertilization and chemical protection), integrated (with reduced nitrogen fertilization and chemical protection), and organic (with no chemical protection and organic fertilizer).
Chemical Analyses:
- AA Analysis: AA content in carrot biomass was measured using HPLC with UV detection. Different farming conditions and seeding densities were examined, and AA content was cultivar-dependent.
- Carotene Analysis: The study focused on β-carotene and α-carotene content in fresh carrot samples using HPLC. The effect of nitrogen fertilization and seeding density on carotene levels was observed.
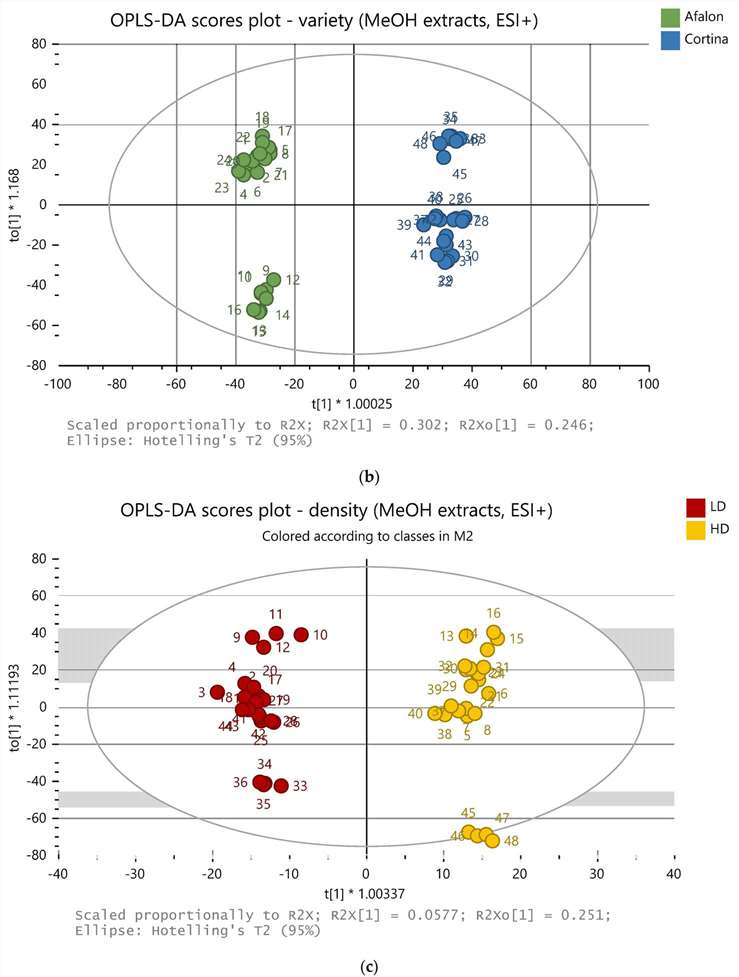
- Metabolomic Fingerprinting: Metabolomic analysis was conducted using UHPLC-Q-TOF-MS. Methanolic and ethylacetate extracts were analyzed in both positive and negative ionization modes. Multivariate data analysis techniques, including OPLS-DA, were used for sample differentiation.
- Chemometric Analysis: Marker compounds were selected based on their importance for sample differentiation. These compounds included saccharides, polyphenolic compounds, and lipids, with various biological activities such as antioxidants, anti-inflammatory, and antibiotic properties.
Results
AA Content: AA levels varied significantly between the two cultivars, with Cortina exhibiting higher AA content. Nitrogen fertilization did not negatively impact AA content in either cultivar.
Carotene Content: Carotene levels (β-carotene and α-carotene) were cultivar-dependent and influenced by farming conditions. An increase in carotene content with higher nitrogen fertilization was observed in Afalon, while seeding density also played a role in carotene synthesis.
Metabolomic Fingerprinting: Metabolomic analysis successfully differentiated carrots based on farming system, cultivar, and seeding density. Various compounds were identified, including those with biological activities such as antioxidants and antimicrobial properties.
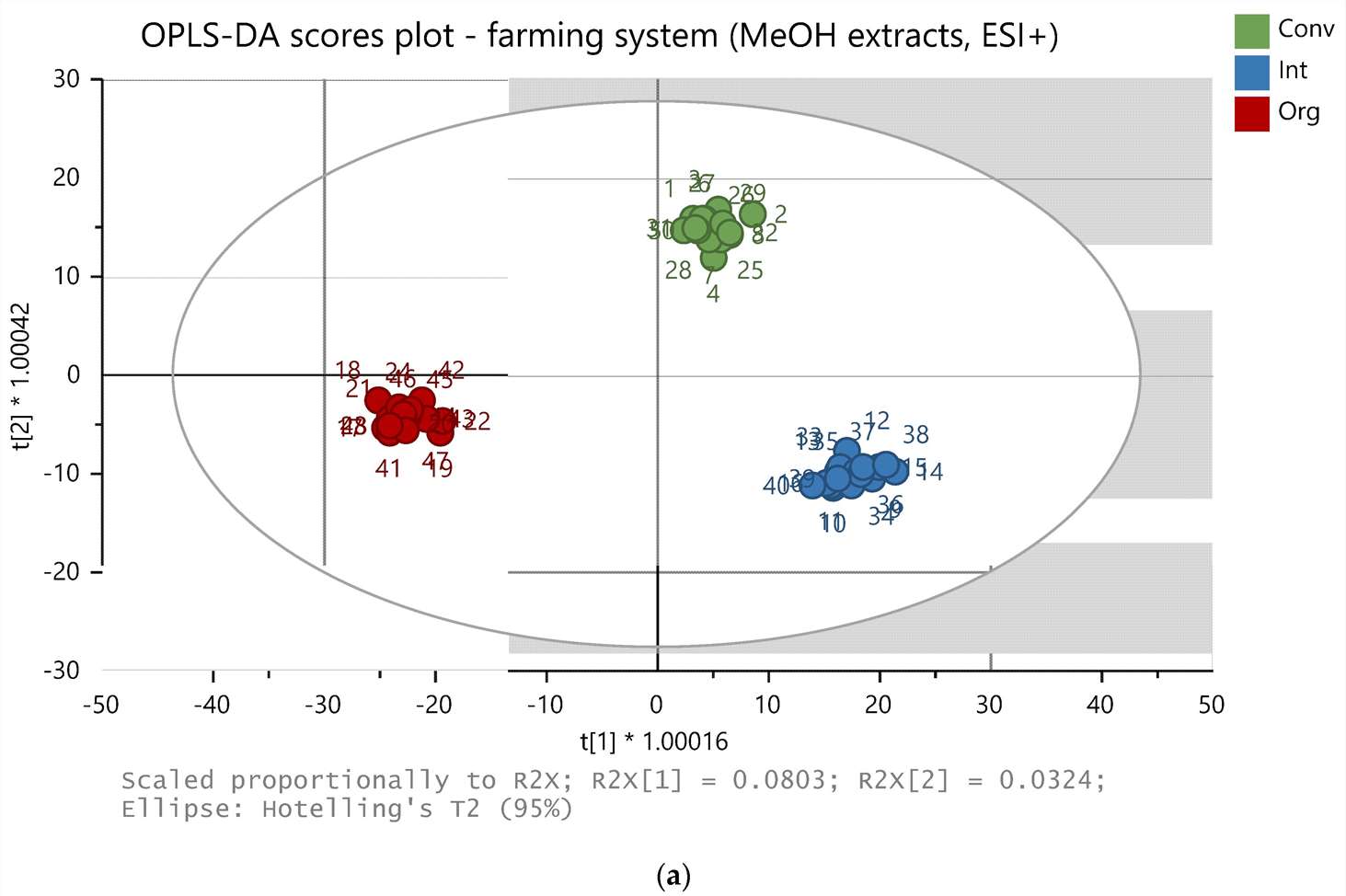 OPLS-DA model of 48 samples (methanolic extracts, positive ion mode).
OPLS-DA model of 48 samples (methanolic extracts, positive ion mode).
Reference
- Schulzova, Vera, et al. "Assessment of Carrot Production System Using Biologically Active Compounds and Metabolomic Fingerprints." Agronomy 12.8 (2022): 1770.


 Workflow for Metabolomics Service
Workflow for Metabolomics Service OPLS-DA model of 48 samples (methanolic extracts, positive ion mode).
OPLS-DA model of 48 samples (methanolic extracts, positive ion mode).