Summary
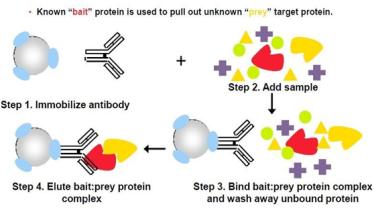
Today we are going to discuss the Co-immunoprecipitation, which we always use to enrich low-abundance protein, including detecting interaction of two target proteins in vivo, defining a new partner of particular proteins and interacting protein complexes in native state. We are familiar with these applications. However, there is still something that you should know before co-immunoprecipitation experiment, such as, its theory, advantages and disadvantages.
| Principle of co-immunoprecipitation experiment | Co-immunoprecipitation, a protocol approach to study protein-protein interactions, is mainly relying on the specificity of antigen and antibody. And it is also the most effective way to detect the physiological interaction of two proteins in complete cells. Some protein-protein interaction will remain intact after lysis under non-denaturing conditions. If antibody X is used to co-immunoprecipitate protein X, so the protein Y, which can combine with X in vivo, will be co-immunoprecipitated. Currently, protein A is widely used to combine and solidify with beads of argarose. And then protein A on beads will be refined after interacting with the solution with antigens and antibodies. |
| Advantages | 1.Proteins that interact in Co-IP are post-translationally modified and conformationally natural. 2.Proteins that interact in Co-IP are in a natural state can be generated. 3.Interacting protein complexes in a natural state can be obtained. |
| Disadvantages | 1.Low-affinity and instantaneous protein-protein interactions may not be detected in Co-IP. 2.Two proteins may not be directly combined. There may be some influence of another substance. 3.In order to select detecting antibody, researchers should predict the target protein. If the forecasting is incorrect, nothing will be obtained. |
Co-immunoprecipitation is a significant method to detect protein-protein interaction. Before this experiment, researchers should pay more attention to these aspects. So the experiment result will be ideal.
Related service: