Sequence Confirmation of DNA & RNA Drugs
Creative Proteomics is committed to providing comprehensive DNA & RNA sequence confirmation services to support your research and applications. Our cutting-edge technologies and highly skilled team ensure the utmost accuracy and reliability of your DNA & RNA sequences.
Introduction of DNA & RNA Drug Sequence Confirmation
DNA & RNA sequence confirmation is a crucial technique employed by the biopharmaceutical industry to ensure the accuracy and integrity of DNA & RNA used in diagnostic and therapeutic applications. Traditional methods like liquid chromatography and electrospray ionization mass spectrometry (LC/ESI-MS) have been well-established for this purpose.
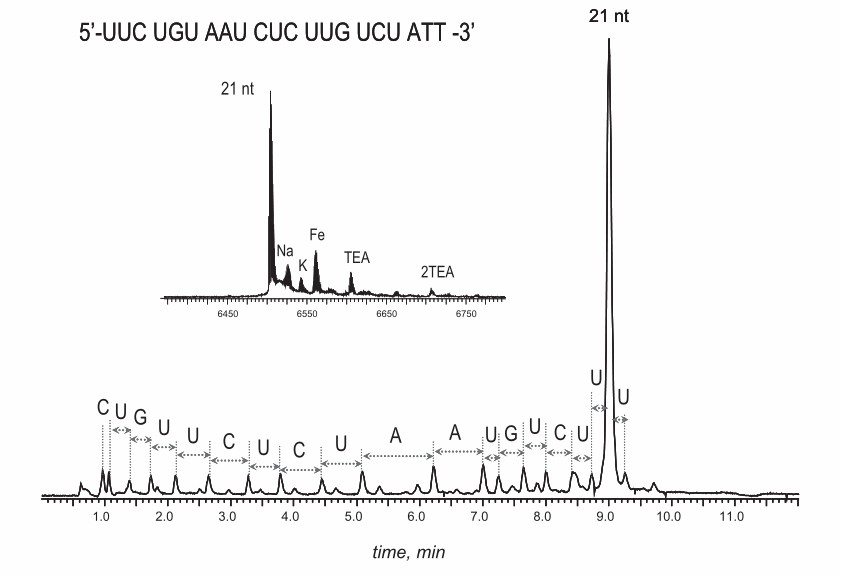 Fig. 1. UPLC/MS of RNA, demonstrating separation of failed sequences and by-products. (Ivleva V, et al., 2010)
Fig. 1. UPLC/MS of RNA, demonstrating separation of failed sequences and by-products. (Ivleva V, et al., 2010)
Our DNA & RNA Drug Sequence Confirmation Services
Creative Proteomics provides professional DNA & RNA sequence confirmation services, our state-of-the-art facilities and team of highly skilled experts enable us to offer a wide range of services for DNA & RNA sequence confirmation. We employ hydrophilic interaction liquid chromatography (HILIC) coupled with tandem mass spectrometry (MS/MS) in negative electrospray ionization (ESI) mode to analyze DNA & RNA with exceptional accuracy and precision. Here are some of the detailed services we can provide in DNA & RNA sequence confirmation.
Mass Spectrometry
Mass spectrometry techniques, like MALDI-TOF (Matrix-Assisted Laser Desorption/Ionization Time-of-Flight), can be employed to analyze the molecular weight and composition of the DNA & RNA, further confirming its identity.
Capillary Electrophoresis
Capillary electrophoresis allows us to analyze the size and purity of the DNA & RNA, helping to identify any impurities or truncated sequences.
Sequence Alignment
We perform sequence alignment to compare your DNA & RNA sequence against a reference sequence or a known database to identify any variations or mutations.
Reporting and Documentation
We furnish detailed reports encompassing sequencing results, analysis findings, and any pertinent recommendations or observations. Our comprehensive documentation not only assures traceability but also facilitates adherence to regulatory requirements if necessary.
Customized Solutions
Our services are tailored to accommodate specific customer requirements, whether it involves batch testing of synthesized DNA & RNAs or undertaking large-scale projects.
Consultation and Support
Our team of experts stands ready to provide guidance and support throughout the entire process, spanning from the design of DNA & RNA sequences to result interpretation and resolution of any potential issues that may arise.
Data Security and Confidentiality
We rigorously uphold protocols for data security and confidentiality, safeguarding your intellectual property and sensitive information.
Through the provision of these comprehensive services, our objective is to guarantee the accuracy, dependability, and quality of DNA & RNAs for a diverse array of applications encompassing research, diagnostics, and therapeutics.
Process of DNA & RNA Drug Sequence Confirmation
Sample Submission
Simply send us your synthesized DNA & RNA samples, and our team will guide you through the submission process, ensuring proper labeling and documentation.
Quality Check
Upon receiving your samples, we conduct a thorough quality check to assess their purity and integrity.
Sequencing Analysis
We employ advanced sequencing technologies to decipher the nucleotide sequence of your DNA & RNAs accurately.
Data Analysis
Our bioinformatics experts then analyze the sequencing data, cross-referencing it with your intended sequence and industry-standard databases.
Validation Report
A comprehensive validation report is generated, detailing the confirmed sequence and any observed modifications or discrepancies, if applicable.
DNA & RNA Drug Sequence Confirmation Application Fields
Our DNA & RNA sequence confirmation service finds application in various areas.
- Assessment of DNA & RNA Drug Candidates.
- Quality Control During the Manufacturing Process.
- Pharmacokinetics and Pharmacodynamics.
- Optimizing Dosage Regimens.
Creative Proteomics is at the forefront of providing high-quality services for DNA & RNA Sequence Confirmation. Our cutting-edge technologies, skilled experts, and advanced data analysis tools empower researchers and pharmaceutical companies to gain precise insights into their DNA & RNA samples, advancing drug development with precision and confidence. If you are interested in our services, please contact us for more detailed information.
Reference
- Ivleva V, Yu Y, Gilar M. (2010). Ultra-performance liquid chromatography/tandem mass spectrometry (UPLC/MS/MS) and UPLC/MS(E) analysis of RNA oligonucleotides. Rapid Commun Mass Spectrom. 24(17):2631-2640.
For research use only, not intended for any clinical use.

 Fig. 1. UPLC/MS of RNA, demonstrating separation of failed sequences and by-products. (Ivleva V, et al., 2010)
Fig. 1. UPLC/MS of RNA, demonstrating separation of failed sequences and by-products. (Ivleva V, et al., 2010)