Protein Complexes Analysis Through 2D Blue Native / Sds-Page
Biological membranes harbor intricate protein complexes that play pivotal roles in diverse cellular functions. Creative Proteomics offers a powerful approach to globally dissect the subunit composition of these protein complexes using the 2D Blue Native / SDS-PAGE analysis method. This technique involves a two-dimensional separation process, with the first dimension utilizing blue native polyacrylamide gel electrophoresis (BN-PAGE) followed by a second dimension separation via SDS-PAGE, creating a comprehensive view of the protein complexes.
High Resolution Analysis with BN-PAGE
BN-PAGE stands as an innovative technology for finely resolving protein complexes. Blue Native PAGE has significant advantages in analyzing protein-protein interactions and membrane protein complexes, with a separation range spanning from 100 kDa to 10 MDa. The blue native electrophoresis protocol not only determines the size and relative abundance of protein complexes but also unveils their subunit constitution. These protein complexes orchestrate cellular and organelle functions across diverse domains, encompassing essential processes such as cell development, transcription, respiration, transport, and metabolism. The dynamic nature and varying stability of protein complexes make their identification and analysis challenging, especially given their potential for partial and temporal changes.
Two-Dimensional Approach in 2D BN/SDS-PAGE
In 2D BN/SDS-PAGE, protein samples or protein complexes undergo native separation in the first-dimension BN-PAGE. Coomassie blue dye, with its negative charge and non-specific protein binding capability, is utilized in this phase. Coomassie blue acts as a stabilizer and preserves complex structures during electrophoresis. This dye also mitigates protein aggregation during the stacking step. The electrophoretic mobility is influenced by the negative charge of the bound Coomassie blue, combined with the size and shape of the complex.
Integration of BN-PAGE and SDS-PAGE
For 2D BN/SDS-PAGE, protein samples or complexes are denatured with SDS after BN-PAGE separation in the gel strip before being introduced to a second-dimension SDS-PAGE gel. The integration of these two techniques provides distinct insights into the samples.
Visualization of Protein Complexes
The 2D BN/SDS-PAGE approach yields a visualization where monomeric proteins migrate diagonally due to the gradient gel setup. Protein complex components reside below this diagonal. Subunits of the same protein complex align vertically in the second dimension, while multiple spots in a horizontal line denote the presence of a protein within various distinct complexes.
Incorporating both BN-PAGE and SDS-PAGE dimensions, 2D BN/SDS-PAGE furnishes comprehensive data on size, composition, stoichiometry, and relative abundance of protein complexes. This method stands as a potent tool for studying intricate protein interactions and structures.
Using 2D BN/SDS-PAGE and mass spectrometry analysis platform, we can assist you in the following applications:
| Applications of BN-PAGE | |
|---|---|
| Complex Structure Analysis | - Analyzing protein complexes in various biological contexts. - Separating protein subunits within complexes to determine composition, size, and abundance. - Studying organelles (e.g., mitochondria, chloroplasts, endoplasmic reticulum) to understand complex assembly and organization. |
| Protein-Protein Interaction Studies | - Preserving native protein interactions for studying protein-protein interactions. - Identifying proteins within the same complex to gain insights into cooperative functions. - Exploring changes in interactions under different cellular conditions. |
| Membrane Protein Complexes | - Studying composition and organization of membrane protein complexes. - Enhancing understanding of cellular processes (e.g., oxidative phosphorylation) involving membrane proteins. - Maintaining complex integrity under native conditions. |
| Comprehensive Characterization | - Combining with mass spectrometry and other tools for identifying and characterizing complex components. - Identifying specific proteins and post-translational modifications for exploring complex functionality. |
| Physiological and Pathological Insights | - Investigating changes in complex composition under diverse physiological and pathological conditions. - Uncovering alterations in complex organization and interactions, providing insights into cellular responses. |
Workflow of 2D BN/SDS-PAGE Mass Spectrometry Analysis Service
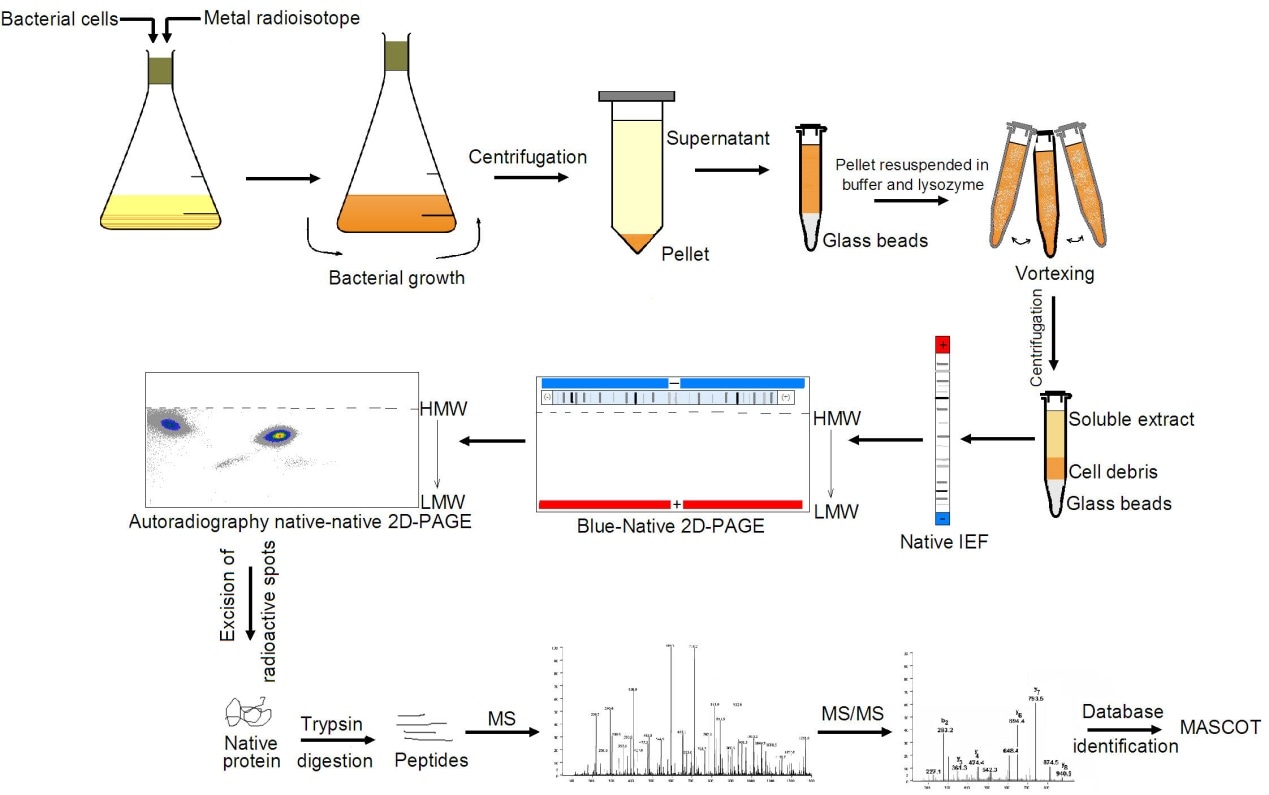
Advantages of Blue Native PAGE (BN-PAGE)
Broad Applicability: BN-PAGE is applicable for separating soluble and insoluble membrane protein complexes, and is not constrained by sample sources.
Efficiency and Reliability: BN-PAGE is characterized by its simplicity, efficiency, and reliability, without the need for expensive equipment.
Resolution of Individual Complexes: Certain proteins may simultaneously exist in multiple complexes, a challenge for methods like immunoprecipitation or yeast two-hybrid. BN-PAGE stands as the sole method capable of discerning individual complexes, holding crucial significance for studying the dynamic regulation of protein complexes.
Application in Whole-Cell Lysates: In recent years, BN-PAGE has evolved to enable the separation of protein complexes from mammalian whole-cell lysates, expanding its applicability in breadth and depth.
Holistic Depiction of Complex Structures: In conjunction with proteomics techniques, BN-PAGE offers a holistic perspective on the composition and interactions of various complex structures within living organisms. This aids in revealing general principles of protein interactions, gradually unraveling the complexities of intracellular regulatory networks, and identifying the protein basis underpinning significant life processes.





