What Is Ames Test
The Ames test is a commonly employed method for assessing the genotoxicity of substances by evaluating their potential harm to human health through the detection of mutagenic properties. This test is characterized by its high sensitivity and good reproducibility.
Principle of the Ames Test
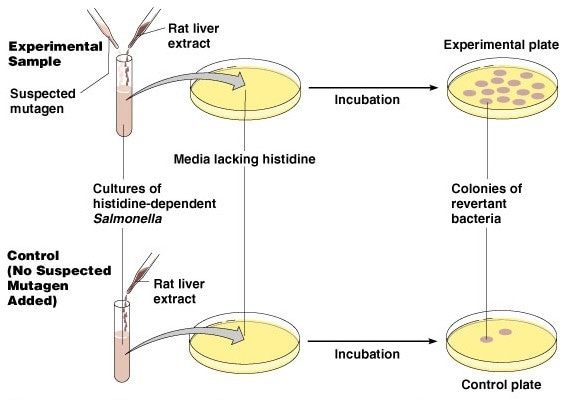
The Ames test, also known as the Bacterial Reverse Mutation Test, is used to detect the mutagenicity and genetic toxicity of chemical compounds. This test utilizes histidine-deficient strains of Salmonella enterica or tryptophan-deficient strains of Escherichia coli, which are unable to synthesize histidine or tryptophan on their own and rely on exogenous sources of these amino acids for growth. These mutant strains cannot survive on selective culture media lacking histidine or tryptophan. In the presence of mutagenic substances, however, these mutants can revert to a wild-type phenotype through reverse mutation, allowing them to grow even in the absence of histidine or tryptophan. Commonly used Salmonella strains include TA98, TA100, TA1535, and TA1537, among others. Each strain is capable of detecting different types of mutagens, and multiple strains can be used simultaneously in a single test, with any one of them exhibiting a reversion considered a positive result.

Ames Testing Procedure
Selection of Appropriate Salmonella Strains: Choose Salmonella strains sensitive to histidine (e.g., TA98, TA100, etc.).
Preparation of Test Medium: Prepare a medium containing essential nutrients and histidine.
Cell Pre-culturing: Cultivate the cells in a medium lacking histidine to induce a low metabolic state.
Cell Treatment: Introduce the test compound into the medium and expose it to Salmonella strains for a defined period.
Plate Spreading: Plate the treated cells onto agar plates containing minimal histidine and other essential nutrients, then incubate.
Counting of Mutant Colonies: Observe the appearance of mutant colonies and count their numbers. If the mutant colony count is significantly higher than the control group, it indicates that the test compound possesses mutagenic activity.
Ames Tester Strains
| Strains | Mutation | Type | Target | rfa | uvr- | pKM101 | pAQ1 |
|---|---|---|---|---|---|---|---|
| Salmonella | TA98 | hisD3052 | Frame-Shift | GCGCGCGC | √ | B | √ |
| TA100 | hisG46 | Base-Pair | GGG | √ | B | √ | |
| TA1535 | hisG46 | Base-Pair | GGG | √ | B | ||
| TA1537 | hisC3076 | Frame-Shift | +1(nearCCC) | √ | B | ||
| TA97(a) | hisD6610 | Frame-Shift | GGGGGG | B | √ | ||
| TA102 | hisG428 | Frame-Shift | A:T | √ | |||
| E. coli | uvrA[pKM 101] | trpE65 | Base-Pair | A:T | A | √ | |
| UvrA | trpE65 | Base-Pair | A:T | A | |||
| pKM101 | trpE65 | Base-Pair | A:T | √ |
rfa: Mutations leading to defects in the cell surface lipopolysaccharide (LPS) layer, making bacteria more permeable to various chemicals. Elimination of the LPS layer renders Salmonella non-pathogenic.
uvr-: Deletion mutations that eliminate precise excision repair mechanisms, allowing more DNA damage to be repaired through error-prone DNA repair mechanisms. Loss of biotin biosynthesis through a mutation in the biotin operon.
pKM101: This R-factor plasmid enhances mutation induced by chemicals and UV through an error-prone recombinational DNA repair pathway. The plasmid also confers ampicillin resistance.
pAQ1: Plasmid derivative of pBR322 that carries the arc gene for the target DNA sequence and has tetracycline resistance.
Please note that these strains have specific mutations and characteristics that make them suitable for detecting different types of mutagens in the Ames test.
Applications of Ames Testing
Ames testing is conducted in various scientific and regulatory contexts:
Compound Screening: It serves as an initial screening step to assess the mutagenic potential of compounds, aiding in early-stage compound selection.
IND-Enabling Studies: In the context of Investigational New Drug (IND) development, Ames testing can be a pivotal component of preclinical safety assessment.
ICH S2(R1) Standard Battery: Ames testing is included as part of Option 1 or Option 2 in the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) S2(R1) guideline, ensuring compliance with global regulatory standards.
REACH Requirements: Under the Registration, Evaluation, Authorisation, and Restriction of Chemicals (REACH) regulation, Ames testing may be required for chemical substance registration.
Annex VII Testing: Ames testing may be prescribed as part of Annex VII testing, depending on the specific regulatory requirements.
Impurity Assessment: When impurities, degradants, or process intermediates are identified as potential mutagens through in silico analysis in accordance with the ICH M7 guideline, Ames testing becomes a necessary component of impurity assessment.
Our Ames Testing Services
Our genetic toxicity screening services, backed by a professional team with extensive practical experience, are designed to provide high-quality data and rapid turnaround times to support the discovery and development of clinical candidate compounds. Our Ames test services find wide application in molecular pharmacology, early-stage drug discovery, chemical compounds, agrochemicals, biocides, cosmetics, food screening, as well as testing environmental water, soil, air, or wastewater. In addition to offering full standard Ames tests, we also provide a range of screening assays at multiple facilities worldwide:
Micro Ames
Ames MPF
Ames II
Mini Ames
Whether your project requires any type of Ames testing, we are here to provide professional support to ensure the most accurate assessment of the genetic toxicity of your compounds.
Advantages of Ames Test Services
High Sensitivity: The Ames test offers high sensitivity, excellent reproducibility, and simplicity in operation, enabling the rapid identification of compounds with mutagenic properties.
High Throughput: It can be conducted in a high-throughput and automated manner, providing advantages such as reduced consumable usage, minimized drug quantities, shortened testing cycles, objective results, and suitability for large-scale drug screening.
Low Compound Usage: With minimal compound usage, it serves as a rapid method for assessing genetic toxicity in early-stage drug development, particularly when dealing with a wide variety of test compounds, limited sample availability, or precious test substances.